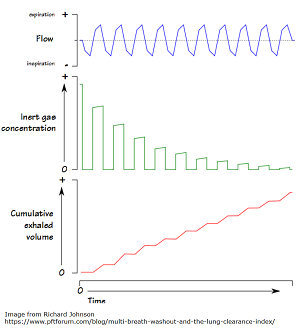
Lung Clearance Index Improved in Children Ages 6-11 on Ivacaftor/Lumacaftor
Ivacaftor/Lumacaftor Improves Lung Clearance Index Score In Children A new phase 3 study, led by The Hospital for Sick Children […]
Outcomes and Tolerances in Cystic Fibrosis Treatment Lumacaftor/Ivacaftor
Last month, the results of a study were published that looked at the progress and side effects of the cystic […]
Orkambi Study On Children Age 6-12 With DDF508: Cystic Fibrosis Treatment
Results are in from a 24-week study of Orkambi (lumacaftor/ivacaftor – a medication used in cystic fibrosis treatment) in 58 children […]
Mom takes on Vertex CEO about pricey Cystic Fibrosis Treatment Drugs
In October, Oklahoma-based journalist and mom Juliana Keeping traveled to Boston with her son to confront Jeffrey Leiden, the CEO of […]